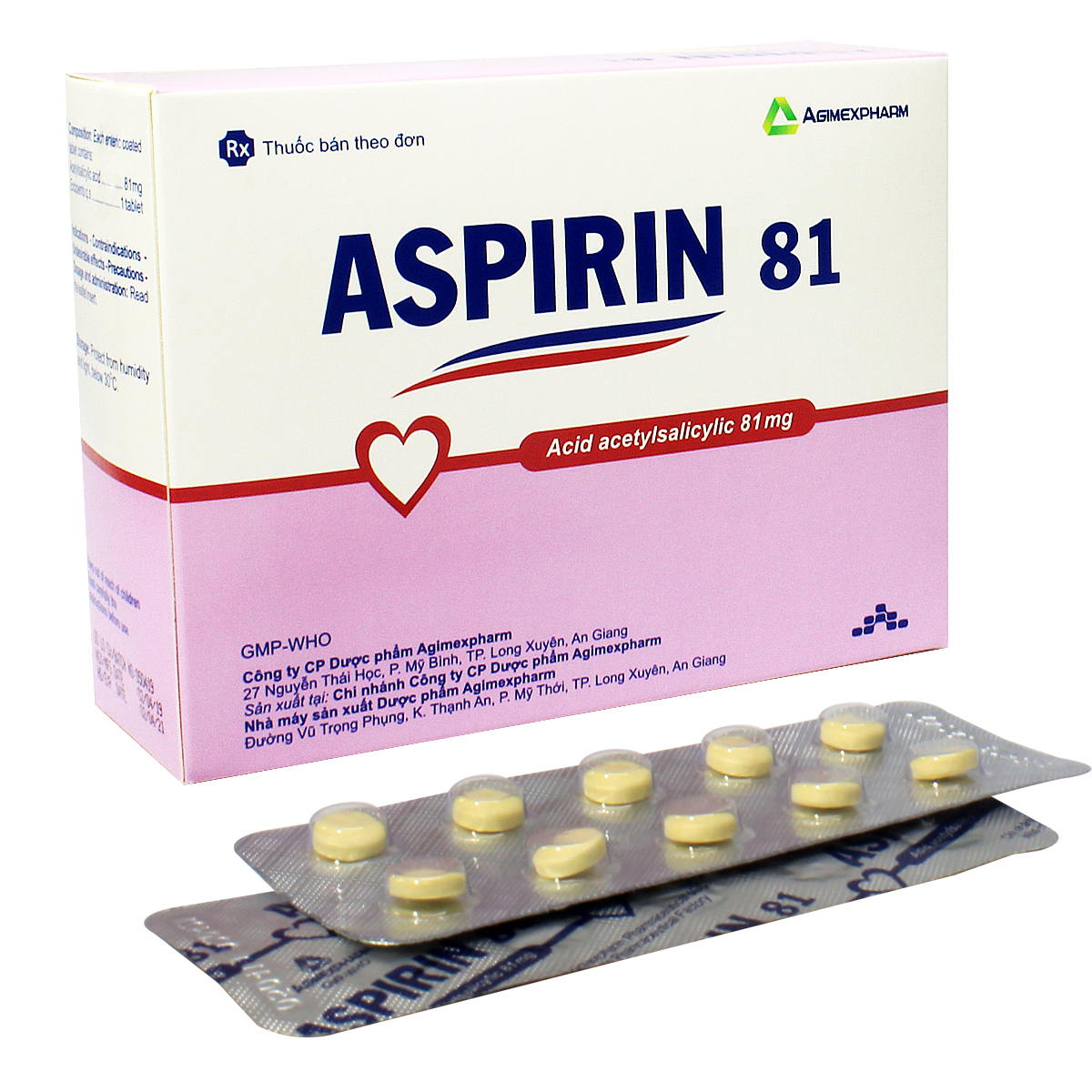
Chỉ định: Phòng ngừa nguyên phát và dự phòng thứ phát nhồi máu cơ tim, đột quỵ.
Liều dùng: Liều thường dùng trong điều trị dài hạn 1 – 2 viên x 1 lần/ngày. Trong một số trường hợp có thể thích hợp với liều cao hơn, đặc biệt trong điều trị ngắn hạn và có thể được sử dụng liều lên đến 4 viên/ngày theo hướng dẫn của bác sỹ.
Cách dùng: Uống nguyên viên thuốc sau khi ăn, không được nhai hay nghiền ra.
Chống chỉ định:
Người mẫn cảm với dẫn xuất salicylat và NSAID.
Người có tiền sử bệnh hen.
Người loét dạ dày – tá tràng.
Tất cả các dạng xuất huyết do thể tạng hay do mắc phải.
Nguy cơ xuất huyết.
Người có tốc độ lọc cầu thận dưới 30ml/phút và xơ gan.
Người mới bị gout.
Dùng liều trên 100mg/ngày cho phụ nữ có thai trong 3 tháng cuối của thai kỳ.
Các trường hợp thận trọng khi dùng thuốc:
Các tình trạng cần thận trọng:
Aspirin hàm lượng 81mg không phù hợp với các chỉ định hạ sốt, chống viêm, giảm đau.
Nên dùng cho bệnh nhân từ 16 tuổi trở lên. Không nên dùng cho bệnh nhân dưới 16 tuổi trừ khi lợi ích mong đợi lớn hơn các nguy cơ. Acid acetylsalicylic có thể là yếu tố góp phần gây ra hội chứng Reye ở trẻ em.
Cần thận trọng trước phẫu thuật, dừng điều trị tạm thời nếu cần thiết. Vì có thể gia tăng nguy cơ chảy máu nghiêm trọng trong hoặc sau phẫu thuật (thậm chí các phẫu thuật nhỏ như nhổ răng).
Không nên dùng trong thời kỳ kinh nguyệt vì có thể làm gia tăng chảy máu kinh nguyệt.
Cần thận trọng trong các trường hợp huyết áp cao và những bệnh nhân có tiền sử loét dạ dày, tá tràng, hoặc xuất huyết hoặc đang điều trị bằng các liệu pháp chống đông máu.
Nên báo cáo với bác sỹ bất cứ triệu chứng chảy máu bất thường nào. Nên ngừng điều trị nếu có xuất huyết hoặc loét đường tiêu hóa.
Nên thận trọng khi dùng acid acetylsalicylic ở bệnh nhân suy thận vừa hoặc chức năng gan suy giảm (chống chỉ định nếu nghiêm trọng), hoặc ở những bệnh nhân bị mất nước bởi vì dùng các thuốc kháng viêm không steroid (NSAIDs) có thể dẫn đến suy giảm chức năng thận.
Acid acetylsalicylic có thể thúc đẩy các cơn co thắt phế quản và các cơn hen suyễn hoặc các phản ứng quá mẫn khác. Yếu tố nguy cơ là bệnh hen suyễn, viêm mũi dị ứng, polyp mũi hoặc bệnh hô hấp mãn tính, người có phản ứng dị ứng với các chất khác (ví dụ: Phản ứng da, mẫn ngứa hoặc mày đay).
Các phản ứng phụ trên da nghiêm trọng, bao gồm hội chứng Steven-Johnsons, ít khi được báo cáo có liên quan đến việc dùng acid acetylsalicylic. Nên dừng ngay việc dùng thuốc khi lần đầu tiên xuất hiện phát ban da, tổn thương niêm mạc, hoặc bất kỳ dấu hiệu nào của phản ứng quá mẫn.
Bệnh nhân lớn tuổi đặc biệt nhạy cảm với tác dụng phụ của các thuốc kháng viêm không steroid (NSAIDs), bao gồm acid acetylsalicylic, đặc biệt là xuất huyết tiêu hóa và thủng đường tiêu hóa có thể gây tử vong. Trường hợp điều trị kéo dài, bệnh nhân nên được xem xét thường xuyên.
Không nên điều trị đồng thời Aspirin 81 với các thuốc làm thay đổi quá trình đông máu (ví dụ: Các thuốc chống đông máu như warfarin, thuốc chống cục máu đông và thuốc chống kết tập tiểu cầu, thuốc kháng viêm và các chất ức chế chọn lọc tái hấp thu serotonin), trừ khi được chỉ định rõ, vì có thể làm tăng nguy cơ xuất huyết. Nếu việc kết hợp là bắt buộc, nên theo dõi chặt chẽ các dấu hiệu xuất huyết.
Nên thận trọng ở những bệnh nhân đang dùng các thuốc khác có thể làm tăng nguy cơ loét, như các corticoid dùng đường uống, các chất ức chế chọn lọc tái hấp thu serotonin và các phức hợp của sắt dùng đường uống.
Cần tránh dùng acid acetylsalicylic ở cuối thai kỳ và thông thường trong thời gian cho con bú.
Dùng Aspirin liều thấp làm giảm bài tiết acid uric. Do đó, những bệnh nhân có xu hướng giảm bài tiết acid uric có thể bị các cơn gout cấp.
Dùng Apirin 81 quá liều có thể làm tăng tác dụng hạ đường huyết của các thuốc hạ đường huyết nhóm sulfonylurê và insulin.
Thành phần tá dược của thuốc này có:
. Màu tartrazin (E 102): Có thể gây phản ứng dị ứng.
. Tinh bột mì: Không nên dùng thuốc này cho bệnh nhân bị dị ứng với lúa mì (khác với bệnh Celiac).
Các khuyến cáo dùng thuốc cho phụ nữ có thai và cho con bú:
Phụ nữ có thai:
Liều thấp (lên đến 100mg/ngày): Các nghiên cứu lâm sàng chỉ ra rằng liều dùng lên đến 100mg/ngày hạn chế dùng trong sản khoa, vì yêu cầu giám sát chuyên môn, cho thấy an toàn.
Liều 100 – 500mg/ngày: Có ít thử nghiệm lâm sàng liên quan đến việc dùng liều trên 100mg/ngày đến 500mg/ngày. Do đó, các khuyến cáo được áp dụng cùng với mức liều từ 500mg/ngày trở lên.
Liều từ 500mg/ngày trở lên:
Tác dụng ức chế tổng hợp prostaglandin có thể gây ảnh hưởng xấu đến thai kỳ và/hoặc sự phát triển của phôi thai/bào thai. Dữ liệu từ các nghiên cứu dịch tễ học cho thấy nguy cơ sẩy thai và khuyết tật tim và thành bụng tăng lên sau khi dùng các chất ức chế tổng hợp prostaglandin trong giai đoạn đầu của thai kỳ. Tỉ lệ chắc chắn xảy ra của khuyết tật tim mạch tăng từ dưới 1% lên đến xấp xỉ 1,5%. Nguy cơ này được cho là tăng theo liều và thời gian điều trị. Trên động vật, việc sử dụng các chất ức chế tổng hợp prostaglandin làm sự mất và chết phôi thai tăng lên trước và sau khi cấy. Ngoài ra, nguy cơ mắc các dị dạng khác, bao gồm dị dạng tim mạch, tăng lên, đã được ghi nhận ở động vật được cho dùng chất ức chế tổng hợp prostaglandin trong giai đoạn hình thành các cơ quan. Trong tam cá nguyệt thứ nhất và thứ hai của thai kỳ, không nên dùng acid acetylsalicylic, trừ khi thật sự cần thiết. Nếu dùng acid acetylsalicylic ở phụ nữ cố gắng thụ thai, hoặc trong tam cá nguyệt thứ nhất và thứ hai của thai kỳ, nên dùng liều thấp và thời gian dùng nên ngắn nhất có thể.
Trong tam cá nguyệt thứ ba của thai kỳ, tất cả các chất ức chế tổng hợp prostaglandin có thể khiến cho thai nhi:
Độc tính tim phổi (với sự đóng sớm của ống động mạch và tăng huyết áp phổi).
Rối loạn chức năng thận, có thể tiến triển đến suy thận kèm thiểu ối.
Khiến cho người mẹ và trẻ sơ sinh ở cuối thai kỳ:
Có thể kéo dài thời gian xuất huyết, do tác động chống kết tập có thể xảy ra ở liều rất thấp.
Ức chế cơn co tử cung dẫn đến trì hoãn hoặc kéo dài sự chuyển dạ.
Do đó, acid acetylsalicylic ở liều 100mg/ngày hoặc cao hơn là chống chỉ định trong tam cá nguyệt thứ ba của thai kỳ.
Phụ nữ cho con bú:
Lượng ít salicylat và các chất chuyển hóa của nó được bài tiết vào sữa mẹ. Vì các tác dụng phụ trên trẻ sơ sinh đến nay vẫn chưa được báo cáo, do đó sử dụng thuốc với liều khuyến cáo trong thời gian ngắn không cần phải ngừng cho con bú.
Tác động của thuốc đến khả năng lái xe và vận hành máy móc:
Chưa có thông tin về ảnh hưởng của thuốc đến việc lái tàu xe và vận hành máy móc.
Tương tác của thuốc với các thuốc khác và các loại tương tác khác:
Tương tác chống chỉ định:
Methotrexat (liều > 15mg/tuần): Sự kết hợp methotrexat và acid acetylsalicylic làm tăng độc tính huyết học của methotrexat do giảm độ thanh thải cầu thận của methotrexat bởi acid acetylsalicylic. Do đó, không dùng đồng thời methotrexat (liều > 15mg/tuần) với Aspirin.
Tương tác nên tránh:
Các thuốc tăng bài tiết acid uric trong nước tiểu, ví dụ: Probenecid
Salicylat làm đảo ngược tác dụng của probenecid, tránh dùng phối hợp.
Tương tác phải thận trọng hoặc xem xét kỹ khi dùng:
Các thuốc chống đông máu, ví dụ: Coumarin, heparin, warfarin: Làm tăng nguy cơ xuất huyết do ức chế chức năng tiểu cầu, làm tổn thương niêm mạc tá tràng và thay đổi vị trí gắn kết với protein huyết tương của các thuốc chống đông máu. Thời gian chảy máu nên được theo dõi.
Các thuốc chống kết tập tiểu cầu (ví dụ: Clopidogrel và dipyridamol) và các thuốc ức chế chọn lọc tái hấp thu serotonin (ví dụ: Sertralin hoặc paroxetin): Tăng nguy cơ xuất huyết đường tiêu hóa.
Các thuốc chống đái tháo đường (ví dụ: Sulfonylurê): Salicylic có thể làm tăng tác dụng hạ đường huyết của sulfonylurê.
Digoxin và lithi: Acid acetylsalicylic làm giảm bài tiết digoxin và lithi, dẫn đến tăng nồng độ trong huyết tương. Nên theo dõi nồng độ digoxin và lithi trong huyết tương khi bắt đầu và khi kết thúc điều trị bằng acid acetylsalicylic. Điều chỉnh liều nếu cần thiết.
Các thuốc lợi tiểu và chống tăng huyết áp: Các thuốc kháng viêm không steroid (NSAIDs) có thể làm giảm tác dụng chống tăng huyết áp của thuốc lợi tiểu và các thuốc chống tăng huyết áp khác. Khi kết hợp các thuốc kháng viêm không steroid (NSAIDs) khác với thuốc ức chế men chuyển làm tăng nguy cơ suy thận cấp. Nguy cơ suy thận cấp do giảm độ lọc cầu thận thông qua giảm tổng hợp prostaglandin thận. Nên bổ sung nước cho bệnh nhân và theo dõi chức năng thận tại thời điểm bắt đầu điều trị.
Các thuốc ức chế carbonic anhydrase (ví dụ: Acetazolamid): Có thể dẫn đến nhiễm toan nghiêm trọng và tăng nhiễm độc hệ thần kinh trung ương.
Các glucocorticoid: Tăng nguy cơ loét và xuất huyết đường tiêu hóa.
Methotrexat (liều < 15mg/tuần): Sự kết hợp methotrexat và acid acetylsalicylic làm tăng độc tính huyết học của methotrexat do giảm độ thanh thải cầu thận của methotrexat bởi acid acetylsalicylic. Nên kiểm tra công thức máu hàng tuần trong suốt những tuần đầu tiên. Tăng cường theo dõi ở những bệnh nhân suy giảm chức năng thận nhẹ và người cao tuổi.
Các thuốc kháng viêm không steroid (NSAIDs) khác: Tăng nguy cơ xuất huyết và loét đường tiêu hóa tác dụng hiệp đồng.
Ibuprofen: Dữ liệu thực nghiệm cho thấy ibuprofen có thể ức chế tác dụng chống kết tập tiểu cầu của aspirin liều thấp khi dùng chung.
Ciclosporin, tacrolimus: Tăng độc tính ở thận của ciclosporin và tacrolimus. Nên theo dõi chức năng thận khi kết hợp.
Các antacid: Tăng bài tiết acid acetylsalicylic do tác dụng kiềm hóa nước tiểu của antacid.
Rượu: Dùng đồng thời rượu và acid acetylsalicylic làm tăng nguy cơ xuất huyết đường tiêu hóa.
Tác dụng không mong muốn:
Thường gặp, ADR >1/100
– Tiêu hóa: Buồn nôn, nôn, khó tiêu, khó chịu ở thượng vị, ợ nóng, đau dạ dày, loét dạ dày – ruột.
– Hệ thần kinh trung ương: Mệt mỏi.
– Da: Ban, mày đay.
– Huyết học: Thiếu máu tan máu.
– Thần kinh – cơ và xương: Yếu cơ.
– Hô hấp: Khó thở.
– Khác: Sốc phản vệ.
Ít gặp, 1/1000 < ADR <1/100
– Hệ thần kinh trung ương: Mất ngủ, bồn chồn, cáu gắt.
– Nội tiết và chuyển hóa: Thiếu sắt.
– Huyết học: Chảy máu ẩn, thời gian chảy máu kéo dài, giảm bạch cầu, giảm tiểu cầu, thiếu máu.
– Gan: Độc hại gan.
– Thận: Suy giảm chức năng thận.
– Hô hấp: Co thắt phế quản.
Hướng dẫn cách xử trí ADR:
ADR trên hệ thần kinh trung ương có thể hồi phục hoàn toàn trong vòng 2 – 3 ngày sau khi ngừng thuốc. Nếu có các triệu chứng chóng mặt, ù tai, giảm thính lực hoặc thương tổn gan, phải ngừng thuốc. Ở người cao tuổi, nên điều trị với liều aspirin thấp nhất có hiệu lực và trong thời gian ngắn nhất có thể được. Điều trị sốc phản vệ do aspirin với liệu pháp giống như khi điều trị các phản ứng phản vệ cấp tính. Adrenalin là thuốc chọn lọc và thường kiểm soát dễ dàng chứng phù mạch và mày đay.
Quá liều và cách xử trí:
Quá liều: Khi uống một lượng lớn acid acetylsalicylic có thể dẫn đến thở sâu, nhanh, ù tai, điếc, giãn mạch, ra mồ hôi.
Cách xử trí:
Làm sạch dạ dày bằng cách gây nôn (chú ý cẩn thận để không hít vào) hoặc rửa dạ dày, cho uống than hoạt. Theo dõi và nâng đỡ các chức năng cần thiết cho sự sống. Điều trị sốt cao; truyền dịch, chất điện giải, hiệu chỉnh mất cân bằng acid – base; điều trị chứng tích ceton; giữ nồng độ Glucose huyết tương thích hợp.
Gây bài niệu bằng cách kiềm hóa nước tiểu để tăng thải trừ salicylat.
Thực hiện truyền thay máu, thẩm tách máu, thẩm tách phúc mạc, nếu cần khi quá liều nghiêm trọng.
Theo dõi phù phổi, co giật và thực hiện liệu pháp thích hợp nếu cần.
Truyền máu hoặc dùng Vitamin K nếu cần để điều trị chảy máu.
Các đặc tính dược lực học:
Acid acetylsalicylic ức chế không thuận nghịch cyclooxygenase, do đó ức chế tổng hợp prostaglandin. Các tế bào có khả năng tổng hợp cyclooxygenase mới sẽ có thể tiếp tục tổng hợp prostaglandin sau khi nồng độ acid salicylic giảm. Tiểu cầu là tế bào không có nhân, không có khả năng tổng hợp cyclooxygenase mới, do đó cyclooxygenase bị ức chế không thuận nghịch, cho tới khi tiểu cầu mới được tạo thành. Như vậy, acid acetylsalicylic ức chế không thuận nghịch kết tập tiểu cầu, cho tới khi tiểu cầu mới được tạo thành.
Acid acetylsalicylic còn ức chế sản sinh prostaglandin ở thận. Sự sản sinh prostaglandin ở thận ít quan trọng về mặt sinh lý với người bệnh có thận bình thường, nhưng có vai trò rất quan trọng trong duy trì lưu thông máu qua thận ở người suy thận mãn tính, suy tim, suy gan hoặc có rối loạn về thể tích huyết tương. Ở những người bệnh này, tác dụng ức chế tổng hợp prostaglandin ở thận của acid acetylsalicylic có thể dẫn đến suy thận cấp tính, giữ nước và suy tim cấp tính.
Các đặc tính dược động học:
Acid acetylsalicylic khi uống được hấp thu nhanh với mức độ cao. Trong khi được hấp thu qua thành ruột, cũng như khi ở gan và máu, acid acetylsalicylic được thủy phân thành acid salicylic, có cùng tác dụng dược lý như acid acetylsalicylic.
Khả dụng sinh học uống: 68% ± 3.
Gắn với protein huyết tương: 49%. Tăng urê máu làm giảm gắn với protein huyết tương.
Độ thanh thải: 9,3 ml/phút/kg ± 1,1. Độ thanh thải thay đổi ở người cao tuổi và người xơ gan.
Thời gian bán thải: 0,25 giờ ± 0,03. Thời gian bán thải thay đổi ở người viêm gan.
Đào thải qua thận chủ yếu dưới dạng acid salicylic tự do và các chất chuyển hóa liên hợp.


 English
English