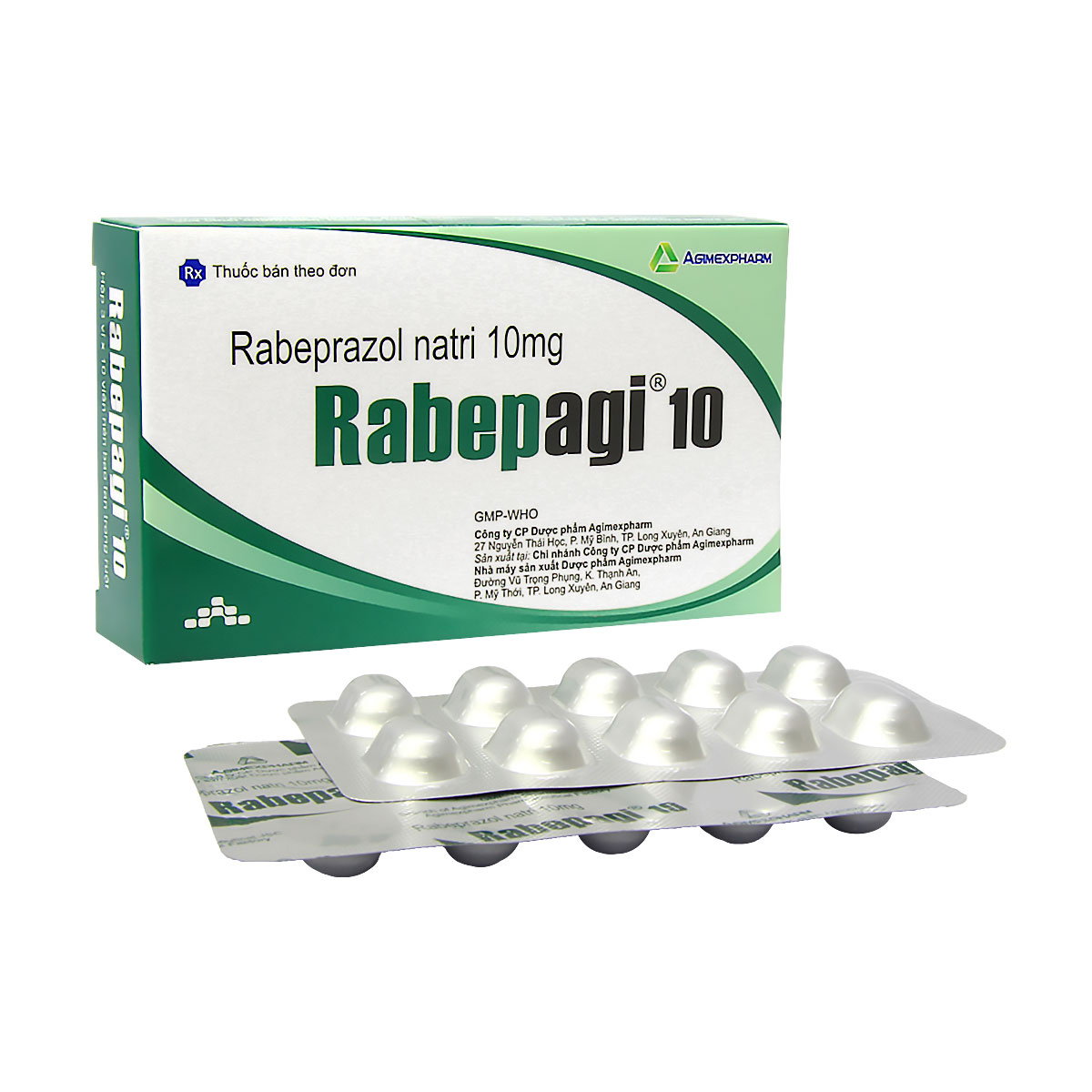
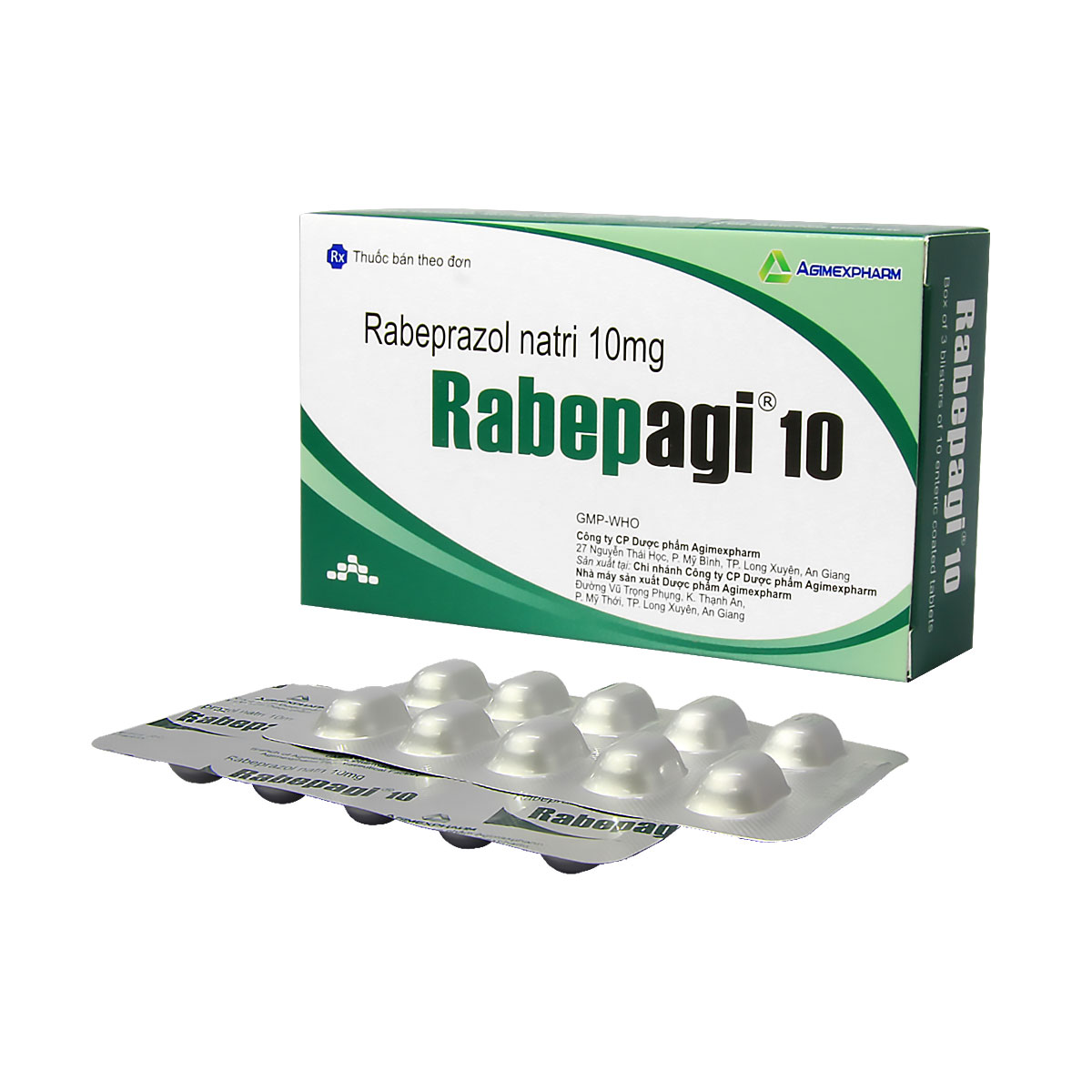
Chỉ định:
- Loét tá tràng cấp tính.
- Loét dạ dày lành tính cấp tính.
- Hội chứng trào ngược dạ dày – thực quản có hoặc không có viêm thực quản, loét hoặc trầy xước.
- Hội chứng Zollinger – Ellison.
- Loét hành tá tràng và loét dạ dày lành tính: Kết hợp với liệu trình kháng sinh thích hợp để diệt Helicobacter pylori.
Cách dùng, liều dùng:
Cách dùng:
Uống thuốc vào buổi sáng, trước bữa ăn, nên uống nguyên viên thuốc, không nhai hoặc bẻ viên.
Liều dùng:
Liều dùng cho người lớn:
- Loét tá tràng cấp tính: 20 mg/lần/ngày, uống trong 4 tuần nếu vết loét chưa liền hoàn toàn.
- Loét dạ dày lành tính cấp tính: 20 mg/lần/ngày, uống trong 6 tuần, tiếp tục thêm 6 tuần nếu vết loét chưa liền hoàn toàn.
- Hội chứng trào ngược dạ dày – thực quản có triệu chứng loét hoặc trầy xước: 20 mg/lần/ngày, uống trong 4 – 8 tuần.
- Bệnh trào ngược dạ dày thực quản phải điều trị lâu dài: Điều trị duy trì, khuyến cáo liều dùng hàng ngày 10 – 20 mg/lần/ngày, phụ thuộc vào đáp ứng của bệnh nhân.
- Điều trị triệu chứng bệnh trào ngược dạ dày – thực quản không viêm thực quản: Liều khuyến cáo 10 mg/lần/ngày cho tới 4 tuần, sau đó 10 mg/lần/ngày khi cần. Nếu không thấy triệu chứng bệnh được kiểm soát trong 4 tuần, bệnh nhân cần được tái khám.
- Hội chứng Zollinger – Ellison: Liều khởi đầu 60 mg/lần/ngày, sau đó có thể tăng đến 100 mg/lần/ngày hay 60 mg x 2 lần/ngày. Liều dùng nên được điều chỉnh tùy theo nhu cầu của mỗi bệnh nhân và dùng liên tục cho đến khi hết triệu chứng lâm sàng.
- Loét hành tá tràng và loét dạ dày lành tính kết hợp với nhiễm pylori: Khuyến cáo kết hợp các thuốc sau đây trong vòng 7 ngày, uống vào buổi sáng và buổi tối: Rabeprazol 20 mg/lần, 2 lần/ngày + clarithromycin 500 mg/lần, 2 lần/ngày và amoxicillin 1g/lần, 2 lần/ngày.
Bệnh nhân suy gan, suy thận: Không cần điều chỉnh liều.
Một số lưu ý đặc biệt về xử lý thuốc trước và sau khi sử dụng thuốc:
Không có yêu cầu đặc biệt về xử lý thuốc sau khi sử dụng.
Chống chỉ định:
Mẫn cảm với rabeprazol, các dẫn chất của benzimidazol hoặc bất kỳ thành phần nào của thuốc.
Trẻ em, do độ an toàn khi sử dụng rabeprazol cho trẻ em chưa được xác định (chưa có các thử nghiệm lâm sàng đầy đủ).
Phụ nữ mang thai hoặc đang cho con bú.
Cảnh báo và thận trọng khi dùng thuốc:
- Trước khi chỉ định cho người bị loét dạ dày dùng rabeprazol, nên loại trừ khả năng bị u ác tính vì thuốc có thể che lấp các triệu chứng và gây trở ngại chẩn đoán bệnh.
- Bệnh nhân rối loạn chức năng gan:
Các bất thường enzym gan đã được thấy trong các thử nghiệm lâm sàng và cũng đã được báo cáo sau khi đưa thuốc ra thị trường. Trong phần lớn các trường hợp không thể xác định được nguyên nhân, các bất thường này không biến chứng và mất đi khi ngừng sử dụng rabeprazol.
- Điều trị với các thuốc ức chế bơm proton (PPI) có thể làm tăng nguy cơ nhiễm khuẩn đường tiêu hóa (ví dụ nhiễm Salmonella, Campylobacter và Clostridium difficile).
- Không thể loại trừ nguy cơ phản ứng quá mẫn chéo với các thuốc ức chế bơm proton khác.
- Rối loạn tạo máu: Đã có báo cáo sau khi đưa thuốc ra thị trường về rối loạn tạo máu (giảm tiểu cầu và giảm bạch cầu trung tính).Trong phần lớn các trường hợp không thể xác định được nguyên nhân, các rối loạn này không biến chứng và mất đi khi ngừng sử dụng rabeprazol.
- Bệnh lupus ban đỏ da bán cấp (SCLE): Thuốc ức chế bơm proton có liên quan đến một số trường hợp bệnh lupus ban đỏ da bán cấp rất hiếm khi xảy ra.Nếu tổn thương xảy ra, đặc biệt là ở vùng da tiếp xúc với ánh nắng mặt trời, và nếu kèm theo đau khớp, bệnh nhân cần nhanh chóng liên hệ với bác sỹ để xem xét ngừng dùng thuốc rabeprazol. Người mắc bệnh lupus ban đỏ da bán cấp sau khi điều trị với thuốc ức chế bơm proton thì có thể tăng nguy cơ mắc bệnh lupus ban đỏ da bán cấp khi điều trị với các thuốc ức chế bơm proton khác.
- Tương tác với warfarin: Tương tác của rabeprazol và warfarin chưa được đánh giá đầy đủ ở bệnh nhân. Đã có báo cáo về tăng INR và thời gian prothrombin ở bệnh nhân dùng đồng thời thuốc ức chế bơm proton và warfarin. Tăng INR và thời gian prothrombin có thể dẫn đến chảy máu bất thường và thậm chí tử vong. Bệnh nhân điều trị đồng thời thuốc ức chế bơm proton và warfarin có thể cần được theo dõi tăng INR và thời gian prothrombin.
- Viêm thận kẽ cấp tính: Viêm thận kẽ cấp tính đã được quan sát ở bệnh nhân dùng thuốc ức chế bơm proton (PPI) bao gồm rabeprazol.Viêm thận kẽ cấp tính có thể xảy ra tại bất kỳ thời điểm nào trong khi điều trị bằng PPI và thường do phản ứng quá mẫn. Ngừng dùng rabeprazol nếu viêm thận kẽ cấp tính phát triển.
- Gãy xương: Một số nghiên cứu quan sát ở người lớn được công bố cho thấy rằng liệu pháp PPI có thể làm tăng nguy cơ gãy xương hông, cổ tay hoặc cột sống liên quan đến loãng xương.Nguy cơ gãy xương tăng ở những bệnh nhân dùng liều cao, tức là dùng liều nhiều ngày, và dùng liệu pháp PPI lâu dài (1 năm hoặc lâu hơn). Bệnh nhân nên dùng liều thấp nhất và thời gian điều trị PPI ngắn nhất phù hợp với tình trạng đang được điều trị. Bệnh nhân có nguy cơ bị gãy xương liên quan đến loãng xương nên được điều trị theo phác đồ điều trị chuẩn.
- Thiếu vitamin B12: Việc điều trị hàng ngày với bất kỳ loại thuốc ức chế acid nào trong một thời gian dài (ví dụ, dài hơn 3 năm) có thể dẫn đến giảm hấp thu cyanocobalamin (vitamin B12) do giảm hoặc thiếu acid trong dịch vị.Chẩn đoán này nên được xem xét nếu các triệu chứng lâm sàng phù hợp với sự thiếu hụt cyanocobalamin được quan sát ở bệnh nhân điều trị bằng rabeprazol.
- Giảm magnesi máu: Tình trạng giảm magnesi máu, có triệu chứng và không có triệu chứng, hiếm khi xảy ra ở bệnh nhân điều trị bằng PPI trong ít nhất 3 tháng, phần lớn là sau một năm điều trị.Các tác dụng phụ nghiêm trọng bao gồm co giật, loạn nhịp và động kinh. Ở hầu hết các bệnh nhân, điều trị hạ magnesi huyết cần phải bổ sung magnesi và ngưng sử dụng PPI.
Đối với bệnh nhân dự kiến sẽ được điều trị lâu dài với thuốc ức chế bơm proton hay trên bệnh nhân đang dùng đồng thời thuốc ức chế bơm proton với digoxin hoặc những thuốc có thể làm hạ magnesi huyết (như thuốc lợi tiểu), thì bác sỹ nên chú ý kiểm tra nồng độ magnesi huyết thanh trước khi bắt đầu kê đơn với thuốc ức chế bơm proton và kiểm tra định kỳ sau đó.
- Tương tác với methotrexat: Y văn cho thấy sử dụng đồng thời PPI với methotrexat (chủ yếu ở liều cao) có thể làm tăng và kéo dài nồng độ methotrexat và/hoặc chất chuyển hóa của nó trong huyết thanh, có thể dẫn đến độc tính của methotrexat.Trong trường hợp điều trị liều cao methotrexat, có thể xem xét tạm thời ngưng dùng thuốc PPI ở một số bệnh nhân.
- Thành phần tá dược của thuốc này có phẩm màu tartrazin (E 102): Có thể gây phản ứng dị ứng.
Sử dụng thuốc cho phụ nữ có thai và cho con bú:
Thời kỳ có thai:
Chống chỉ định với phụ nữ có thai do chưa có các dữ liệu nghiên cứu về độ an toàn của rabeprazol đối với phụ nữ trong thời kỳ mang thai.
Phụ nữ có khả năng mang thai hoặc đang sử dụng các biện pháp tránh thai nên tham khảo ý kiến bác sỹ cẩn thận trước khi điều trị với thuốc này.
Không có thông tin về độc tính trên thai nhi.
Thời kỳ cho con bú:
Chống chỉ định với bà mẹ trong thời kỳ cho con bú do chưa xác định được rabeprazol có bài tiết qua sữa mẹ hay không và cũng chưa có nghiên cứu nào được tiến hành trên các bà mẹ cho con bú.
Ảnh hưởng của thuốc lên khả năng lái xe, vận hành máy móc:
Chưa có bằng chứng về ảnh hưởng của thuốc lên khả năng lái xe, vận hành máy móc. Tuy nhiên thận trọng đối với người đang lái xe và vận hành máy móc do thuốc có thể gây đau đầu, chóng mặt, buồn ngủ.
Tương tác, tương kỵ của thuốc:
Tương tác của thuốc với các thuốc khác và các loại tương tác khác:
- Với ketoconazol hoặc itraconazol: Có thể giảm sự hấp thu ketoconazol hoặc itraconazol. Vì vậy nếu dùng chung với rabeprazol cần giám sát điều chỉnh liều ketoconazol hoặc itraconazol.
- Tránh dùng đồng thời rabeprazol với: Erlotinib, nelfinavir, delavirdin, posaconazol.
- Rabeprazol có thể làm giảm nồng độ/tác dụng của atazanavir, clopidogrel, dabigatran, etexilat, dasatinib, erlotinib, indinavir, muối sắt, itraconazol, ketoconazol, mesalamin, mycophenolat, nelfinavir.
- Rabeprazol có thể làm tăng nồng độ/tác dụng của các thuốc là cơ chất CYP2C19 và CYP2C8 (mức độ rủi ro cao) methotrexat, saquinavir, voriconazol.
- Methotrexat:
Các trường hợp báo cáo, các nghiên cứu dược động học đã công bố và các phân tích hồi cứu cho thấy dùng đồng thời PPI và methotrexat với liều cao (xem thông tin kê toa methotrexat) có thể làm tăng và kéo dài lượng methotrexat và/hoặc chất chuyển hóa hydroxymethotrexat trong huyết thanh. Tuy nhiên, không có nghiên cứu chính thức về tương tác thuốc của methotrexat với PPI đã được tiến hành (xem Cảnh báo và thận trọng khi dùng thuốc).
Tương kỵ của thuốc:
Do không có các nghiên cứu về tính tương kỵ của thuốc, không trộn lẫn thuốc này với các thuốc khác.
Tác dụng không mong muốn
Các phản ứng có hại được phân nhóm theo tần suất: Rất thường gặp (ADR ≥ 1/10), thường gặp (1/100 ≤ ADR < 1/10), ít gặp (1/1.000 ≤ ADR < 1/100), hiếm gặp (1/10.000 ≤ ADR < 1/1.000), rất hiếm gặp (ADR < 1/10.000); không thể ước lượng tần suất được liệt kê “Chưa rõ tần suất”.
Các tác dụng phụ thường gặp với rabeprazol trong các thử nghiệm lâm sàng có kiểm soát là nhức đầu, tiêu chảy, đau bụng, suy nhược, đầy hơi, phát ban và khô miệng. Đa số các tác dụng phụ bất lợi xảy ra trong các nghiên cứu lâm sàng đều có mức độ nhẹ hoặc trung bình và có tính chất thoáng qua.
Các phản ứng phụ sau đây đã được báo cáo từ thử nghiệm lâm sàng và kinh nghiệm sau khi tiếp thị.
Thường gặp
Nhiễm trùng và nhiễm ký sinh trùng: Nhiễm trùng.
Rối loạn tâm thần: Mất ngủ.
Rối loạn hệ thần kinh: Nhức đầu, chóng mặt.
Rối loạn hô hấp, lồng ngực và trung thất: Ho, viêm họng, viêm mũi.
Rối loạn tiêu hóa: Tiêu chảy, nôn, buồn nôn, đau bụng, táo bón, đầy hơi.
Rối loạn cơ xương và mô liên kết: Đau không rõ nguyên nhân, đau lưng.
Rối loạn tổng quát: Suy nhược, hội chứng giống cúm.
Ít gặp
Rối loạn tâm thần: Lo lắng, bồn chồn.
Rối loạn hệ thần kinh: Buồn ngủ.
Rối loạn hô hấp, lồng ngực và trung thất: Viêm phế quản, viêm xoang.
Rối loạn tiêu hóa: Chứng khó tiêu, khô miệng, ợ hơi.
Rối loạn da và mô dưới da: Phát ban, ban đỏ.
Rối loạn cơ xương và mô liên kết: Đau cơ, chứng chuột rút, đau khớp, gãy xương hông, cổ tay hoặc cột sống.
Rối loạn thận và tiết niệu: Nhiễm trùng đường tiết niệu.
Rối loạn tổng quát: Đau ngực, ớn lạnh, sốt.
Xét nghiệm: Tăng men gan.
Hiếm gặp
Rối loạn tâm thần: Trầm cảm.
Rối loạn máu và hệ bạch huyết: Tăng hoặc giảm bạch cầu, giảm bạch cầu trung tính, giảm tiểu cầu.
Rối loạn hệ thống miễn dịch: Phản ứng dị ứng cấp tính (ví dụ sưng mặt, hạ huyết áp và khó thở).
Rối loạn chuyển hóa và dinh dưỡng: Biếng ăn.
Rối loạn mắt: Rối loạn thị giác.
Rối loạn tiêu hóa: Viêm dạ dày, viêm miệng, rối loạn vị giác.
Rối loạn gan mật: Viêm gan, vàng da, bệnh não gan.
Rối loạn da và mô dưới da: Ngứa, ra mồ hôi, phản ứng bỏng rộp.
Rối loạn thận và tiết niệu: Viêm thận kẽ.
Khác: Tăng cân.
Rất hiếm gặp
Rối loạn da và mô dưới da: Hồng ban đa dạng, hội chứng hoại tử da nhiễm độc (TEN), hội chứng Stevens-Johnson (SJS).
Chưa rõ tần suất
Rối loạn tâm thần: Nhầm lẫn.
Rối loạn chuyển hóa và dinh dưỡng: Hạ natri trong máu, hạ magnesi trong máu.
Rối loạn mạch máu: Phù ngoại biên.
Rối loạn da và mô dưới da: Bệnh lupus ban đỏ dưới da bán cấp.
Rối loạn vú và hệ thống sinh sản: To vú đàn ông.
Hướng dẫn cách xử trí ADR
Phải ngừng thuốc khi có biểu hiện tác dụng không mong muốn nặng.
Quá liều và cách xử trí
Quá liều:
Cho đến nay chưa có kinh nghiệm về trường hợp dùng quá liều rabeprazol do cố ý hoặc vô tình. Trường hợp quá liều tối đa được xác nhận không vượt quá 60 mg hai lần mỗi ngày hoặc 160 mg một lần mỗi ngày. Ảnh hưởng thường là nhẹ, biểu hiện giống như các tác dụng phụ đã biết và có thể hồi phục mà không có sự can thiệp y tế.
Xử trí: Chưa có thuốc giải độc đặc hiệu.
Do rabeprazol kết hợp rất mạnh với protein nên không thể thải trừ bằng phương pháp thẩm phân. Trong trường hợp quá liều, cần điều trị các triệu chứng và điều trị hỗ trợ.
Tích cực theo dõi để có biện pháp xử trí kịp thời.
Đặc tính dược lực học:
Nhóm dược lý: Thuốc ức chế bơm proton.
Mã ATC: A02BC04
Rabeprazol là dẫn chất benzimidazol, có tác dụng ức chế bơm proton.
Rabeprazol có tác dụng ức chế tiết dịch vị cả cơ bản và trong tình trạng kích thích, không có tính chất kháng acetylcholin hoặc đối kháng thụ thể histamin H2, bằng cách ức chế men H+/K+ ATPase ở tế bào thành của niêm mạc dạ dày. Enzym này được coi là bơm acid, hydrogen hoặc proton trong tế bào thành nên rabeprazol được coi là thuốc ức chế bơm proton. Rabeprazol được gắn vào enzym này ngăn chặn giai đoạn cuối cùng của sự tiết dịch vị. Trong tế bào thành của dạ dày, rabeprazol được proton hóa và chuyển thành sulfenamid hoạt động và sau đó gắn với cystein của bơm proton làm enzym này bất hoạt.
Đặc tính dược động học:
Hấp thu: Rabeprazol được hấp thu nhanh chóng và nồng độ đỉnh trong huyết tương đạt được sau khi uống 3,5 giờ. Sinh khả dụng đường uống khoảng 52% do bị chuyển hóa qua gan lần đầu và không thay đổi nhiều khi dùng liều một lần hay liều lặp lại.
Phân phối: Rabeprazol gắn kết khoảng 97% với protein huyết tương.
Chuyển hóa: Thuốc được chuyển hóa mạnh qua gan bởi hệ thống isoenzym cytochrom P450 (CYP2C19 và CYP3A4) thành các dẫn chất thioether, thioether của acid carboxylic, sulfon và desmethylthioether.
Thải trừ: Thời gian bán thải trong huyết tương khoảng 1 giờ, tăng gấp 2 đến 3 lần ở bệnh nhân suy gan, gấp 1,6 lần ở những người enzym CYP2C19 chuyển hóa chậm và tăng 30% ở người cao tuổi.
Các chất chuyển hóa được bài tiết chủ yếu qua nước tiểu (khoảng 90%), phần còn lại thải trừ qua phân.


 English
English