| Dosage and administration | Dosage: |
|---|---|
| Contraindications | Hypersensitivity to esomeprazole, other proton pump inhibitors or any excipient of this medicine. |
| Warnings and precautions for use | Before starting esomeprazole therapy, the doctor must rule out the possibility of gastric cancer because the medicine can mask the symptoms, and so delay the diagnosis of cancer. |
| Recommendation for pregnancy and breastfeeding | Pregnancy |
| Effects on ability to drive and use machines | There is no evidence to prove esomeprazole has influence on the ability to drive or use machines. However, it should be noted that the medicine can cause headache, dizziness, drowsiness, visual disturbances. |
| Interactions, incompatibilities of medicine | Effects of esomeprazole on the pharmacokinetics of other medicinal products: Protease inhibitors: Omeprazole has been reported to interact with some protease inhibitors. The clinical importance and the mechanisms behind these reported interactions are not always known. Increased gastric pH during omeprazole treatment may change the absorption of the protease inhibitors. Other possible interaction mechanisms are via inhibition of CYP 2C19. |
| Undesirable effects (ADRs) | In general, esomeprazole is well tolerated when used for both short and long time. |
| Overdose and management | – Overdose: There have been no report on the overdose of esomeprazole in human. |
| Pharmacodynamic properties | Esomeprazole is the S-isomer of omeprazole, and similar to omeprazole it is used in the treatment of gastric and duodenal ulcers, gastroesophageal reflux disease and Zollinger – Ellison syndrome. |
| Pharmacokinetic properties | After oral administration esomeprazole is rapidly absorbed, with peak plasma levels occurring approximately 1-2 hours after dosing. The bioavailability of esomeprazole increases proportionally when the dose is increased and used repeatedly, 68% with dose of 20mg and 89% with dose of 40mg. Absorption of esomeprazole is decreased and delayed if taken with food. After administration of a single 40mg dose of esomeprazole with meal the AUC is decreased by 33% to 53% compared to fasting conditions. Therefore, esomeprazole should be taken at least one hour before meals. Esomeprazole is bound to plasma proteins by about 97%. The apparent volume of distribution at steady state in healthy volunteers is approximately 16 liters. |
| Storage conditions, shelf-life, quality specification of the medicine | Storage conditions: Protect from humidity and light, below 30 degrees C. |
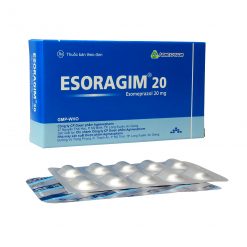
ESORAGIM®40
Composition:Each enteric coated tablet contains:
Esomeprazole magnesium trihydrate equivalent to Esomeprazole. . . . . . . . . . . . .40 mg
Presentation:
Alu-Alu blister containing 10 tablets, 3 blisters in a box.
Indications:
Adults: Esoragim 40 is used for the treatment of:
Gastric and duodenal ulcers.
Severe gastroesophageal reflux disease (erosive esophagitis, ulcers or narrowing (stricture) of the esophagus determined by endoscopy).
Prevention of gastric and duodenal ulcers associated with NSAID therapy.
In combination with appropriate antibacterial therapeutic regimens for the eradication of Helicobacter pylori
Pathological hypersecretory conditions including Zollinger-Ellison Syndrome
Children from 12 years of age and over:
This medicine is indicated for the treatment of gastroesophageal reflux disease with esophagitis.
Sản phẩm tương tự
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
Uncategorized
ESORAGIM 40
Thành phần: Mỗi viên nén bao phim tan trong ruột chứa:
| Esomeprazol (dưới dạng Esomeprazol magnesi trihydrat) | 40 mg |
Quy cách đóng gói:
Hộp 3 vỉ x 10 viên nén bao phim tan trong ruột
Chỉ định:
Người lớn: Thuốc này chỉ định cho các trường hợp sau:
Loét dạ dày – tá tràng.
Bệnh trào ngược dạ dày – thực quản nặng (viêm thực quản trợt xước, loét hoặc thắt hẹp được xác định bằng nội soi).
Dự phòng loét dạ dày – tá tràng do dùng thuốc chống viêm không steroid.
Phối hợp với liệu pháp hợp lý khác để diệt Helicobacter pylori.
Bệnh lý tăng tiết acid bao gồm hội chứng Zollinger – Ellison.
Trẻ em từ 12 tuổi trở lên:
Thuốc này dùng điều trị bệnh trào ngược dạ dày – thực quản có viêm thực quản.
Cách dùng:
Uống thuốc ít nhất 1 giờ trước bữa ăn và nên nuốt nguyên viên thuốc với nước, không được nhai hay nghiền nát.
Liều dùng:
Liều dùng cho người lớn:
Điều trị bệnh trào ngược dạ dày – thực quản nặng có viêm thực quản: 1 viên, 1 lần/ngày, trong thời gian từ 4 đến 8 tuần, có thể uống thêm 4 đến 8 tuần nữa nếu tổn thương chưa liền. Trường hợp nặng có thể tăng liều lên 80 mg/ngày chia 2 lần.
Điều trị loét dạ dày – tá tràng do nhiễm Helicobacter pylori (phối hợp với liệu pháp hợp lý khác): Esomeprazol là một thành phần trong phác đồ điều trị cùng với kháng sinh, ví dụ phác đồ 3 thuốc (cùng với amoxicilin và clarithromycin), liều thường dùng là 1 viên, 1 lần/ngày trong 10 ngày, (Amoxicillin 1g/lần x 2 lần/ngày và clarithromycin 500 mg/lần x 2 lần/ngày trong 10 ngày).
Dự phòng loét dạ dày ở người có nguy cơ cao về biến chứng ở dạ dày – tá tràng, nhưng có yêu cầu phải tiếp tục điều trị bằng thuốc chống viêm không steroid: 1 viên, 1 lần/ngày.
Điều trị hội chứng Zollinger – Ellison: Tùy theo từng cá thể và mức độ tăng tiết acid của dịch dạ dày. Liều khởi đầu khuyến cáo là 1 viên/lần x 2 lần/ngày. Sau đó điều chỉnh liều khi cần thiết. Đa số người bệnh có thể kiểm soát được bệnh ở liều 80-160 mg mỗi ngày. Với liều trên 80 mg mỗi ngày, liều dùng nên được chia 2 lần/ ngày.
Đối tượng đặc biệt:
Bệnh nhân suy gan: Ở bệnh nhân suy gan nặng không dùng quá liều 20 mg/ngày. Đối với bệnh nhân suy gan nhẹ và trung bình không cần giảm liều.
Người suy thận và người cao tuổi: Không cần điều chỉnh liều.
Liều dùng cho trẻ em ≥ 12 tuổi:
Điều trị bệnh trào ngược dạ dày – thực quản có viêm thực quản: 1 viên, 1 lần/ngày trong 4 tuần, có thể uống thêm 4 tuần nữa nếu viêm thực quản chưa lành hoặc vẫn còn triệu chứng.
Chống chỉ định
Quá mẫn với esomeprazol, các thuốc ức chế bơm proton hoặc với bất kỳ thành phần nào của thuốc.
Không phối hợp với nelfinavir, atazanavir.
Các trường hợp thận trọng khi dùng thuốc:
Các tình trạng cần thận trọng:
Trước khi cho người bệnh dùng esomeprazol phải loại trừ khả năng ung thư dạ dày vì thuốc có thể che lấp các triệu chứng, làm chậm chẩn đoán ung thư.
Thận trọng khi dùng ở người bị bệnh gan, người mang thai hoặc cho con bú.
Thận trọng khi dùng cho trẻ em dưới 18 tuổi vì độ an toàn và hiệu quả chưa được xác định.
Thận trọng với những người bệnh dùng esomeprazol dài hạn (đặc biệt là những người điều trị hơn một năm) do có thể gặp những nguy cơ sau:
Viêm teo dạ dày.
Nhiễm khuẩn đường tiêu hóa: Điều trị bằng thuốc ức chế bơm proton có thể làm tăng nguy cơ nhiễm khuẩn đường tiêu hóa (ví dụ nhiễm Salmonella, Campylobacter); có thể tăng nguy cơ tiêu chảy do Clostridium difficile.
Nguy cơ gãy xương: Thuốc ức chế bơm proton, đặc biệt nếu dùng liều cao và thời gian dài (≥ 1 năm), có thể làm tăng nguy cơ gãy xương chậu, xương cổ tay hoặc cột sống do loãng xương. Cơ chế của hiện tượng này chưa được giải thích, nhưng có thể do giảm hấp thu calci không hòa tan do tăng pH dạ dày. Khuyến cáo dùng liều thấp nhất có tác dụng trong thời gian ngắn nhất có thể, phù hợp với tình trạng lâm sàng. Những bệnh nhân có nguy cơ gãy xương do loãng xương nên dùng đủ calci và vitamin D, đánh giá tình trạng xương và quản lý theo hướng dẫn.
Viêm thận kẽ đã được quan sát thấy ở bệnh nhân dùng thuốc ức chế bơm proton bao gồm esomeprazol. Viêm thận kẽ có thể xảy ra ở bất kỳ điểm nào trong khi điều trị PPI và thường là do một phản ứng quá mẫn vô căn. Ngừng esomeprazol nếu viêm thận kẽ cấp tính phát triển
Giảm magnesi trong máu nghiêm trọng đã được báo cáo ở những bệnh nhân được điều trị bằng thuốc ức chế bơm proton (PPI) như esomeprazol trong ít nhất ba tháng, và phần lớn các trường hợp điều trị trong một năm. Biểu hiện nghiêm trọng của giảm magnesi trong máu như mệt mỏi, co cứng cơ, mê sảng, co giật, chóng mặt và rối loạn nhịp thất có thể xảy ra nhưng những biểu hiện này có thể bắt đầu âm thầm và bị bỏ qua. Ở hầu hết các bệnh nhân bị ảnh hưởng, giảm magnesi trong máu được cải thiện sau khi bổ sung magnesi và ngừng PPI.
Giảm hấp thụ vitamin B12: Như tất cả các loại thuốc ức chế acid, esomeprazol có thể làm giảm sự hấp thu vitamin B12 (cyanocobalamin) do giảm hoặc thiếu acid dịch vị. Điều này cần được xem xét ở những bệnh nhân giảm dự trữ trong cơ thể hoặc có yếu tố nguy cơ bị giảm hấp thụ vitamin B12 khi điều trị lâu dài.
Các khuyến cáo dùng thuốc cho phụ nữ có thai và cho con bú:
Thời kỳ mang thai:
Chưa có nghiên cứu đầy đủ khi dùng esomeprazol ở người mang thai. Các nghiên cứu về esomeprazol trên súc vật không cho thấy thuốc có tác động có hại trực tiếp hay gián tiếp trên sự phát triển của phôi/thai. Tuy nhiên, chỉ sử dụng esomeprazol khi thật cần thiết trong thời kỳ mang thai.
Thời kỳ cho con bú:
Chưa biết esomeprazol có bài tiết vào sữa người hay không. Tuy nhiên đã đo được nồng độ của omeprazol trong sữa của phụ nữ sau khi uống 20mg omeprazol.
Esomeprazol có khả năng gây ra các tác dụng không mong muốn nghiêm trọng ở trẻ bú mẹ, vì vậy phải quyết định ngừng cho con bú hoặc ngừng thuốc, tùy theo tầm quan trọng của việc dùng thuốc đối với người mẹ.
Tác động của thuốc đến khả năng lái xe và vận hành máy móc:
Chưa ghi nhận thuốc có tác động nào ảnh hưởng đến người lái xe và vận hành máy móc, tuy nhiên nên lưu ý thuốc có thể gây tác dụng phụ đau đầu, chóng mặt, buồn ngủ, rối loạn thị giác.
Tương tác của thuốc với các thuốc khác và các loại tương tác khác:
Ảnh hưởng của esomeprazol trên dược động học của các thuốc khác:
Các chất ức chế protease: Đã có báo cáo rằng omeprazol tương tác với một số chất ức chế men protease. Chưa rõ tầm quan trọng về lâm sàng và cơ chế tác động của các tương tác đã được ghi nhận. Tăng độ pH dạ dày trong quá trình điều trị với omeprazol có thể dẫn tới thay đổi sự hấp thu của chất ức chế men protease. Cơ chế tương tác khác có thể xảy ra là thông qua sự ức chế enzym CYP2C19.
Atazanavir và nelfinavir: Giảm nồng độ các thuốc này trong huyết thanh đã được ghi nhận khi dùng chung với omeprazol, do đó không khuyến cáo dùng đồng thời các thuốc này. Ở người tình nguyện khỏe mạnh, sử dụng đồng thời omeprazol (40mg, 1 lần/ngày) với atazanavir 300mg/ritonavir 100mg làm giảm đáng kể nồng độ và thời gian tiếp xúc atazanavir (giảm AUC, Cmax và Cmin khoảng 75%). Tăng liều atazanavir đến 400mg đã không bù trừ tác động của omeprazol trên nồng độ và thời gian tiếp xúc atazanavir.
Dùng phối hợp omeprazol (20mg, 1 lần/ngày) với atazanavir 400mg/ritonavir 100mg ở người tình nguyện khỏe mạnh làm giảm khoảng 30% nồng độ và thời gian tiếp xúc atazanavir khi so sánh với nồng độ và thời gian tiếp xúc ghi nhận trong trường hợp dùng atazanavir 300mg/ritonavir 100mg, 1 lần/ngày mà không dùng omeprazol 20mg, 1 lần/ngày. Dùng phối hợp với omeprazol (40mg,1 lần/ngày) làm giảm trị số trung bình AUC, Cmax và Cmin của nelfinavir khoảng 36-39% và giảm khoảng 75-92% trị số trung bình AUC, Cmax và Cmin của chất chuyển hóa hoạt tính có tác động dược lý M8.
Do tác động dược lực và các đặc tính dược động học tương tự của omeprazol và esomeprazol, khuyến cáo không sử dụng esomeprazol đồng thời với atazanavir và chống chỉ định sử dụng esomeprazol đồng thời với nelfinavir.
Tacrolimus: Việc sử dụng đồng thời với esomeprazol đã được báo cáo làm tăng nồng độ huyết thanh của tacrolimus. Cần tăng cường giám sát nồng độ tacrolimus cũng như chức năng thận (độ thanh thải creatinin) và điều chỉnh liều tacrolimus nếu cần thiết.
Methotrexat: Khi dùng đồng thời với các PPI, nồng độ methotrexat được báo cáo là tăng lên ở một số bệnh nhân. Khi dùng liều cao methotrexat, nên cân nhắc dừng tạm thời esomeprazol.
Những thuốc hấp thu phụ thuộc độ pH: Tình trạng giảm độ acid dạ dày khi điều trị bằng esomeprazol và các PPI khác có thể làm giảm hay tăng sự hấp thu của các thuốc khác có cơ chế hấp thu phụ thuộc pH dạ dày. Giống như các thuốc làm giảm độ acid dạ dày khác, sự hấp thu của các thuốc như ketoconazol, itraconazol và erlotinib có thể giảm và sự hấp thu của digoxin có thể tăng lên trong khi điều trị với esomeprazol. Dùng đồng thời omeprazol (20mg/ngày) và digoxin ở các đối tượng khỏe mạnh làm tăng sinh khả dụng của digoxin khoảng 10% (lên đến 30% ở 2 trên 10 đối tượng nghiên cứu). Hiếm có các báo cáo về độc tính của digoxin. Tuy nhiên, cần thận trọng khi dùng esomeprazol liều cao ở bệnh nhân cao tuổi. Cần tăng cường theo dõi điều trị bằng digoxin.
Những thuốc chuyển hóa qua CYP2C19: Esomeprazol ức chế CYP2C19, men chính chuyển hóa esomeprazol. Do vậy, khi esomeprazol được dùng chung với các thuốc chuyển hóa bằng CYP2C19 như diazepam, citalopram, imipramin, clomipramin, phenytoin…, nồng độ các thuốc này trong huyết tương có thể tăng và cần giảm liều dùng. Điều này cần được đặc biệt chú ý khi kê toa esomeprazol theo chế độ điều trị khi cần thiết. Dùng đồng thời với esomeprazol 30mg làm giảm 45% độ thanh thải diazepam (một cơ chất của CYP2C19). Khi dùng đồng thời với esomeprazol 40mg làm tăng 13% nồng độ đáy (trough) của phenytoin trong huyết tương ở bệnh nhân động kinh. Nên theo dõi nồng độ phenytoin trong huyết tương khi bắt đầu hay ngưng điều trị với esomeprazol.
Warfarin: Tăng INR và thời gian prothrombin khi dùng warfarin đồng thời với esomeprazol, có thể gây chảy máu bất thường và tử vong. Theo dõi INR và thời gian prothrombin khi dùng đồng thời esomeprazol với warfarin hoặc các dẫn chất khác của coumarin.
Clopidogrel: Esomeprazol ức chế CYP2C19 nên có thể làm giảm tác dụng của clopidogrel do gây tương tác dược động học với clopidogrel (làm giảm nồng độ trong huyết tương của chất chuyển hóa có hoạt tính của clopidogrel) và tương tác dược lực học với clopidogrel (làm giảm tác dụng kháng tiểu cầu).
Voriconazol: Dùng đồng thời esomeprazol với voriconazol có thể làm tăng tiếp xúc với esomeprazol hơn gấp 2 lần, xem xét ở những bệnh nhân dùng liều cao esomeprazol (240 mg/ngày) như khi điều trị hội chứng Zollinger-Ellison.
Cilostazol: Dùng đồng thời esomeprazol với cilostazol làm tăng nồng độ cilostazol và chất chuyển hóa có hoạt tính của nó, xem xét giảm liều cilostazol.
Cisaprid: Ở người tình nguyện khỏe mạnh, khi dùng chung với 40mg esomeprazol với cisaprid, diện tích dưới đường cong biểu diễn nồng độ cisaprid trong huyết tương theo thời gian (AUC) tăng lên 32% và thời gian bán thải (T1/2) cisaprid kéo dài thêm 31% nhưng nồng độ đỉnh cisaprid trong huyết tương tăng lên không đáng kể. Khoảng QTc hơi kéo dài sau khi dùng cisaprid riêng lẻ, không bị kéo dài hơn nữa khi dùng đồng thời cisaprid với esomeprazol.
Ảnh hưởng của các thuốc khác trên dược động học của esomeprazol:
Thuốc ức chế CYP2C19 và / hoặc CYP3A4: Esomeprazol được chuyển hóa bởi CYP2C19 và CYP3A4. Khi dùng đồng thời esomeprazol với một chất ức chế CYP3A4, clarithromycin (500mg, 2 lần/ngày) làm tăng gấp đôi diện tích dưới đường cong (AUC) của esomeprazol. Dùng đồng thời esomeprazol cùng với một chất ức chế cả hai CYP2C19 và CYP3A4 có thể làm tăng hơn hai lần nồng độ và thời gian tiếp xúc của esomeprazol. Không cần điều chỉnh liều esomeprazol thường xuyên trong những tình huống này. Tuy nhiên, sự điều chỉnh liều cần được lưu ý ở những bệnh nhân suy gan nặng hoặc điều trị lâu dài.
Thuốc cảm ứng CYP2C19 và / hoặc CYP3A4: Các thuốc cảm ứng CYP2C19 hoặc CYP3A4 hoặc cả hai (như rifampicin) có thể gây giảm nồng độ esomeprazol huyết thanh do tăng chuyển hóa esomeprazol.
Tác dụng không mong muốn (ADR):
Nhìn chung, esomeprazol dung nạp tốt cả khi sử dụng trong thời gian ngắn hoặc thời gian dài.
Thường gặp, ADR > 1/100:
- Toàn thân: Đau đầu, chóng mặt, ban ngoài da.
- Tiêu hóa: Buồn nôn, nôn, đau bụng, tiêu chảy, táo bón, đầy hơi, khô miệng.
Ít gặp, 1/1000 < ADR < 1/100:
- Toàn thân: Mệt mỏi, mất ngủ, buồn ngủ, phát ban, ngứa.
- Rối loạn thị giác.
Hiếm gặp, ADR < 1/1000
- Toàn thân: Sốt, đổ mồ hôi, phù ngoại biên, mẫn cảm với ánh sáng, phản ứng quá mẫn (mày đay, phù mạch, co thắt phế quản, sốc phản vệ).
- Thần kinh trung ương: Kích động, trầm cảm, lú lẫn có hồi phục, ảo giác ở người bệnh nặng.
- Huyết học: Chứng mất bạch cầu hạt, giảm bạch cầu, giảm tiểu cầu.
- Gan: Tăng enzym gan, viêm gan, vàng da, suy chức năng gan.
- Tiêu hóa: Rối loạn vị giác.
- Cơ xương: Đau khớp, đau cơ.
- Tiết niệu: Viêm thận kẽ.
- Da: Ban bọng nước, hội chứng Stevens-Johnson, hoại tử biểu bì nhiễm độc, viêm da.
Do làm giảm độ acid của dạ dày, các thuốc ức chế bơm proton có thể làm tăng nguy cơ nhiễm khuẩn ở đường tiêu hóa.
Hướng dẫn cách xử trí ADR: Phải ngừng thuốc khi có biểu hiện tác dụng không mong muốn nặng.
Quá liều và cách xử trí:
- Quá liều: Chưa có báo cáo về quá liều esomeprazol ở người.
- Xử trí: Không có thuốc giải độc đặc hiệu cho esomeprazol. Chủ yếu điều trị triệu chứng và hỗ trợ. Thẩm tách máu không có tác dụng tăng thải trừ thuốc vì esomeprazol gắn nhiều vào protein huyết tương.
Các đặc tính dược lực học:
Esomeprazol là dạng đồng phân S của omeprazol, được dùng tương tự như omeprazol trong điều trị loét dạ dày – tá tràng, bệnh trào ngược dạ dày – thực quản và hội chứng Zollinger – Ellison.
Esomeprazol là một chất ức chế bơm proton làm giảm sự bài tiết acid dạ dày bằng cách gắn với H+/K+-ATPase (còn gọi là bơm proton) ở tế bào thành của dạ dày, làm bất hoạt hệ thống enzym này, ngăn cản bước cuối cùng của sự bài tiết acid hydrocloric vào lòng dạ dày. Vì vậy esomeprazol có tác dụng ức chế dạ dày tiết lượng acid cơ bản và cả khi bị kích thích do bất kỳ tác nhân nào. Thuốc tác dụng mạnh kéo dài.
Tác động lên sự tiết acid dịch vị
Sau khi dùng liều uống esomeprazol 20mg và 40mg, thuốc khởi phát tác động trong vòng 1 giờ. Sau khi dùng lặp lại liều esomeprazol 20mg, 1 lần/ngày trong 5 ngày, sự tiết acid tối đa trung bình sau khi kích thích bằng pentagastrin giảm 90% khi đo ở thời điểm 6-7 giờ sau khi dùng thuốc ở ngày thứ 5.
Sau 5 ngày dùng liều uống esomeprazol 20mg và 40mg, độ pH trong dạ dày > 4 đã được duy trì trong thời gian trung bình tương ứng là 13 và 17 giờ trong vòng 24 giờ ở bệnh nhân trào ngược dạ dày thực quản có triệu chứng. Tỷ lệ bệnh nhân duy trì độ pH trong dạ dày > 4 tối thiểu trong 8, 12 và 16 giờ tương ứng là 76%, 54% và 24% đối với esomeprazol 20mg và 97%, 92% và 56% đối với esomeprazol 40 mg.
Khi dùng AUC như là một tham số đại diện cho nồng độ thuốc trong huyết tương, người ta đã chứng minh được có mối liên hệ giữa sự ức chế tiết acid với nồng độ thuốc và thời gian tiếp xúc.
Các đặc tính dược động học:
Esomeprazol được hấp thu nhanh sau khi uống, đạt nồng độ cao nhất trong huyết tương sau 1-2 giờ. Sinh khả dụng của esomeprazol tăng lên theo liều dùng và khi dùng nhắc lại, đạt khoảng 68% khi dùng liều 20 mg và 89% khi dùng liều 40mg. Thức ăn làm chậm và làm giảm hấp thu esomeprazol, diện tích dưới đường cong (AUC) sau khi uống 1 liều duy nhất esomeprazol 40 mg vào bữa ăn so với lúc đói giảm từ 33% đến 53%. Do đó phải uống esomeprazol ít nhất 1 giờ trước bữa ăn. Khoảng 97% esomeprazol gắn vào protein huyết tương. Thể tích phân bố khi nồng độ thuốc ổn định ở người tình nguyện khỏe mạnh là 16 lít.
Thuốc chuyển hóa chủ yếu ở gan nhờ isoenzym CYP2C19, hệ enzym cytochrom P 450 thành các chất chuyển hóa hydroxy và desmethyl không còn hoạt tính. Phần còn lại được chuyển hóa qua isoenzym CYP3A4 thành esomeprazol sulfon. Khi dùng nhắc lại, chuyển hóa bước đầu qua gan và độ thanh thải của thuốc giảm, có thể do isoenzym CYP2C19 bị ức chế. Tuy nhiên, không có hiện tượng tích lũy thuốc khi dùng mỗi ngày 1 lần.
Thời gian bán thải trong huyết tương khoảng 1,3 giờ. Khoảng 80% liều uống được thải trừ dưới dạng các chất chuyển hóa không có hoạt tính trong nước tiểu, phần còn lại được thải trừ trong phân. Dưới 1% chất mẹ được thải trừ trong nước tiểu.
Các nhóm bệnh nhân đặc biệt
Người chuyển hóa kém qua enzym CYP2C19:
Khoảng 2,9 ± 1,5% bệnh nhân không có men chức năng CYP2C19 và được gọi là nhóm người chuyển hóa kém. Ở các cá nhân này, sự chuyển hóa của esomeprazol có lẽ chủ yếu được xúc tác bởi CYP3A4. Sau khi dùng liều lặp lại esomeprazol 40mg, 1 lần/ngày, AUC trung bình ở người chuyển hóa kém cao hơn khoảng 100% so với bệnh nhân có men chức năng CYP2C19 (nhóm người chuyển hóa mạnh). Nồng độ đỉnh trung bình trong huyết tương tăng khoảng 60%. Những ghi nhận này không ảnh hưởng liều dùng esomeprazol.
Người cao tuổi:
Sự chuyển hóa của esomeprazol không thay đổi đáng kể ở bệnh nhân cao tuổi (71-80 tuổi).
Người suy giảm chức năng gan:
Sự chuyển hóa của esomeprazol có thể bị suy giảm ở bệnh nhân rối loạn chức năng gan từ nhẹ đến trung bình. Tốc độ chuyển hóa giảm ở bệnh nhân rối loạn chức năng gan nặng, dẫn đến làm tăng gấp đôi AUC của esomeprazol. Vì vậy, không dùng quá liều tối đa 20mg ở bệnh nhân rối loạn chức năng gan nặng. Esomeprazol hoặc các chất chuyển hóa chính không có khuynh hướng tích lũy khi dùng 1 lần/ngày.
Người suy giảm chức năng thận:
Chưa có nghiên cứu nào được thực hiện trên bệnh nhân giảm chức năng thận. Vì thận chịu trách nhiệm trong việc bài tiết các chất chuyển hóa của esomeprazol nhưng không chịu trách nhiệm cho sự đào thải thuốc dưới dạng không đổi, người ta cho là sự chuyển hóa của esomeprazol không thay đổi ở bệnh nhân suy giảm chức năng thận.
Trẻ em từ 12-18 tuổi:
Sau khi sử dụng liều lặp lại 20mg và 40mg esomeprazol, tổng nồng độ và thời gian tiếp xúc (AUC) và thời gian đạt nồng độ tối đa trong huyết tương (tmax) ở trẻ 12-18 tuổi tương tự như ở người lớn đối với cả hai liều esomeprazol.
Sản phẩm tương tự
Uncategorized
Tiêu hóa & chuyển hóa
Hóa dược
Tiêu hóa & chuyển hóa
Tiêu hóa & chuyển hóa
Hóa dược
Uncategorized
Hóa dược
Hóa dược
Tiêu hóa & chuyển hóa
Tiêu hóa & chuyển hóa
Hóa dược
Hóa dược
Hóa dược
Tiêu hóa & chuyển hóa
Tiêu hóa & chuyển hóa
Hóa dược
Hóa dược
Tiêu hóa & chuyển hóa
Tiêu hóa & chuyển hóa
Tiêu hóa & chuyển hóa
Hóa dược
Hóa dược
Hóa dược
Hóa dược
Hóa dược
Hóa dược
Uncategorized
Tiêu hóa & chuyển hóa
Hóa dược


 English
English